BPT
Innovations in modern medicine have brought many artificial joints and bionic body parts to patients. Although these devices have been used for years, a prosthetic replacement for the meniscus – the tissue pad located between the thigh and shin bones – is not yet available. Now two clinical trials underway across the U.S. may help make the “artificial meniscus” a reality, providing a new treatment option for millions of Americans with persistent knee pain caused by injured or deteriorated meniscus.
The meniscus is highly vulnerable to tearing, both from physical activity and excess weight. Once damaged, it has a very limited ability to heal. More than 1 million partial meniscectomies to remove or repair a torn meniscus are performed in the U.S. every year – about the same as the total number of hip and knee replacement surgeries combined. However, many patients still experience persistent knee pain following meniscus surgery.
New York area volunteer firefighter Tracy Cullum was diagnosed with a meniscus tear eight years ago, when the 42-year-old mother of two was experiencing a constant ache and the inability to put any weight on her left leg. Although she underwent a partial meniscectomy to treat the tear, her knee pain became progressively worse and resulted in a constant limp while walking.
“I went from working out seven days week to having to stop any exercise activities,” Cullum says. “And my work as a part-time volunteer firefighter was challenging, since it was difficult to move any equipment and my mobility was compromised when responding to an emergency scene.”
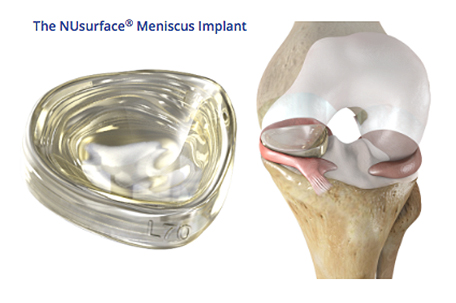
With her activity severely limited, Cullum sought out options to treat her persistent knee pain and discovered that a hospital near her home town is participating in the clinical trial of the NUsurface Meniscus Implant – the first “artificial meniscus” designed to replace the damaged one for patients like Cullum with persistent knee pain due to injured or deteriorated meniscus cartilage.
“There are limited options for patients who experience persistent knee pain following meniscus surgery,” says Dr. Richard Alfred, orthopedic surgeon at Capital Region Orthopaedic Associates and VENUS study investigator. “It is our hope that the NUsurface implant alleviates pain in these patients, allowing them to return to their previous levels of activity.”
Cullum received the implant in July 2016 through a small incision in her knee. She completed a six-week rehabilitation program, and she has now returned to daily leisure activities like hot yoga and her work as a volunteer firefighter.
“In the six months before receiving the NUsurface Meniscus Implant, my knee pain had peaked to where I had a constant dull ache and a limp while walking,” Cullum says. “Now my range of motion is improving, and I am ecstatic to be able to resume one of my favorite activities – volunteering together with my husband and children at our local fire department.”
The NUsurface Meniscus Implant, made of medical-grade plastic, is an investigational device that is inserted into the knee in a minimally invasive procedure through a small incision. It has been used in Europe since 2008 and Israel since 2011.
“It is important to note that NUsurface is an investigational device that is limited by United States law to investigational use,” Dr. Alfred says. “Responses to the NUsurface can and do vary with each patient, so those interested in the trial should discuss their individual situations with a clinical trial physician.”
To be eligible for the clinical studies, you must be between the ages of 30 and 75, have pain after medial meniscus surgery and have had meniscus surgery at least six months ago. To find a study site near you, visit www.activeimplants.com/kneepaintrial.














Leave a Comment